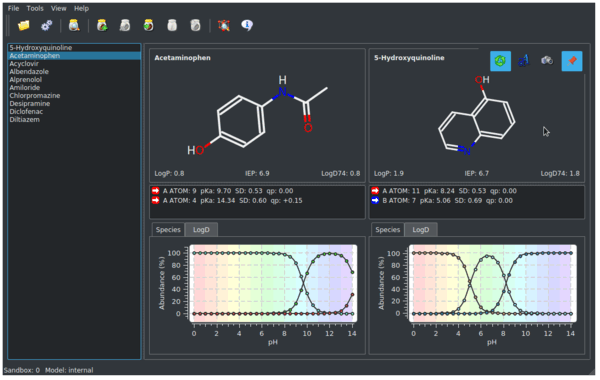
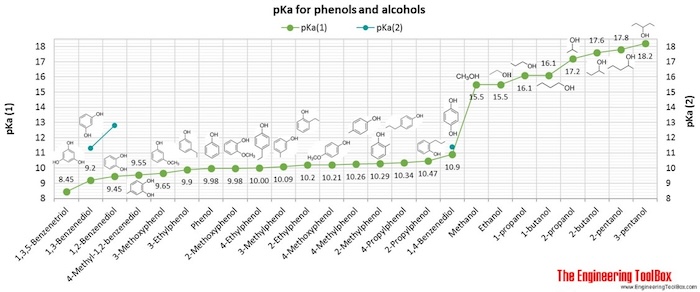
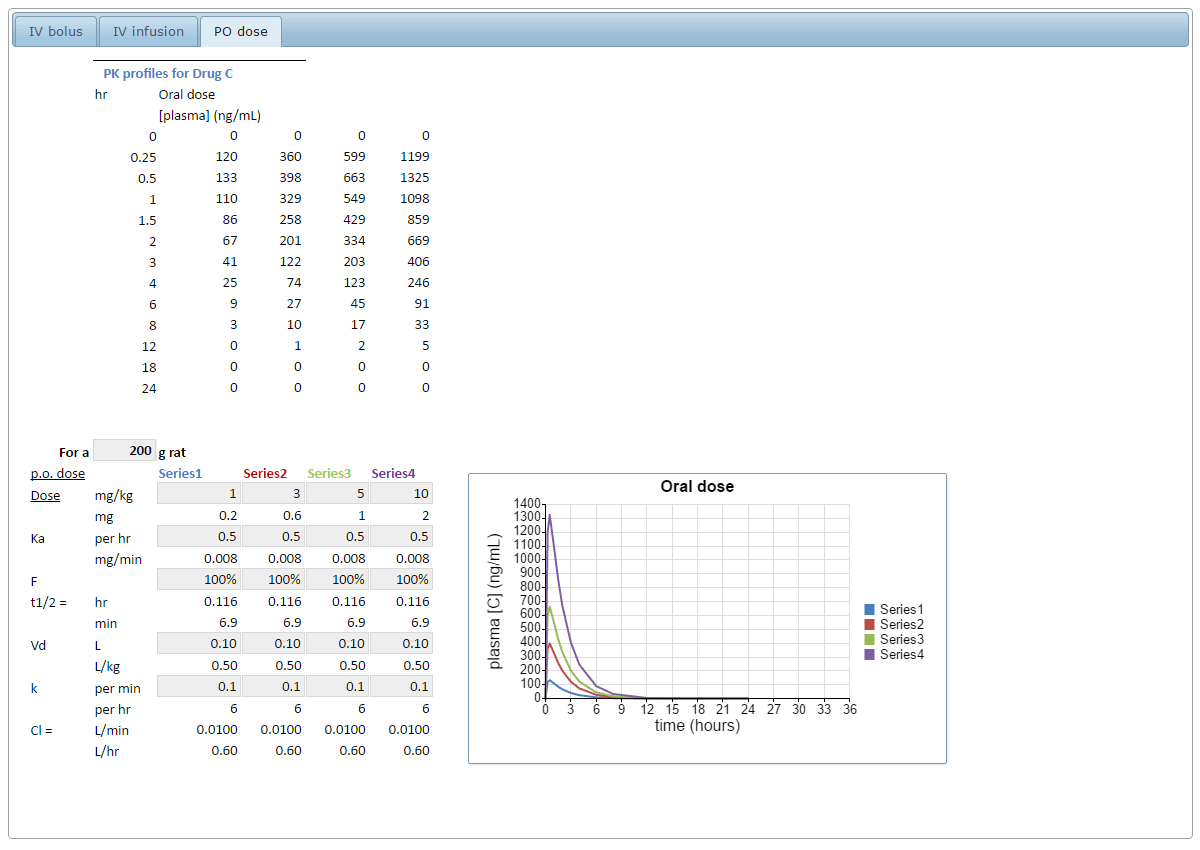
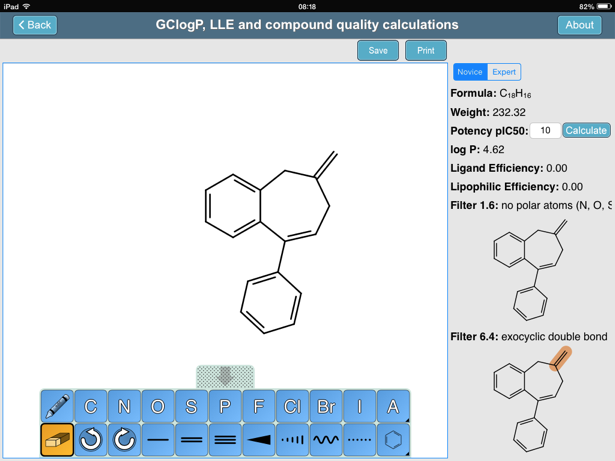
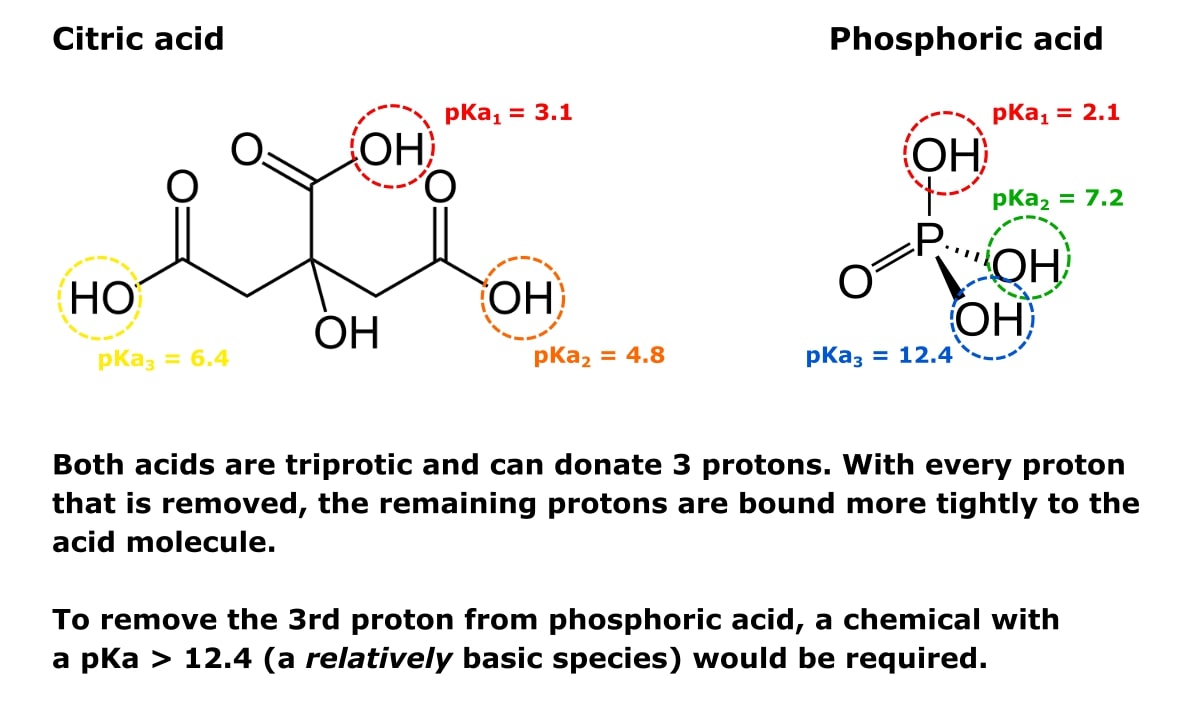
SOLVED: 6) a) Calculate pKa value from the following Ka: Nitromethane, Ka = 5.0 x 10-11 b) What is the Ka of a solution with the pKa = 3.75. 7) Using pKa

pH calculations and more in fundamentals of pharmaceutics. : Calculate pH of 100 ml buffer solution containing 0.1 g acetic acid and 0.2 g sodium actetate.

Ionization and structural properties of mRNA lipid nanoparticles influence expression in intramuscular and intravascular administration | Communications Biology

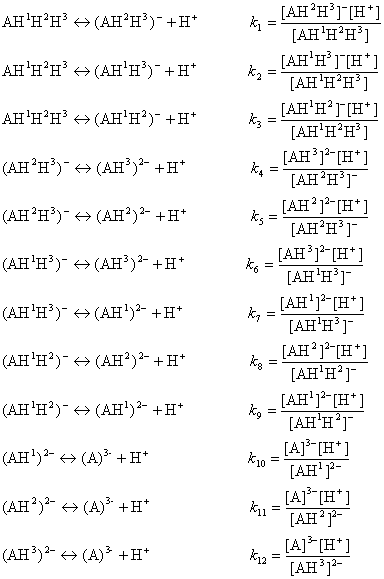



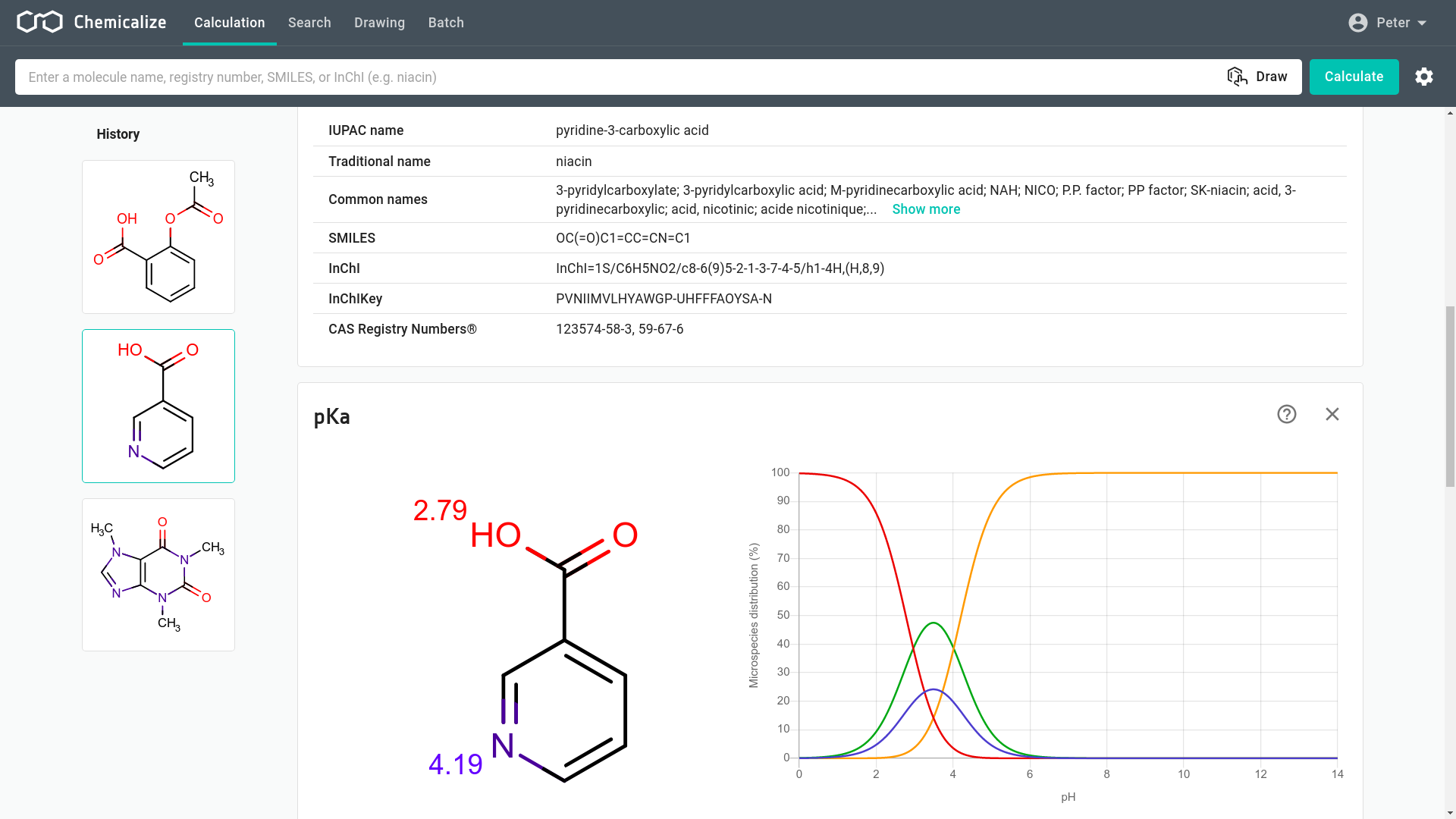
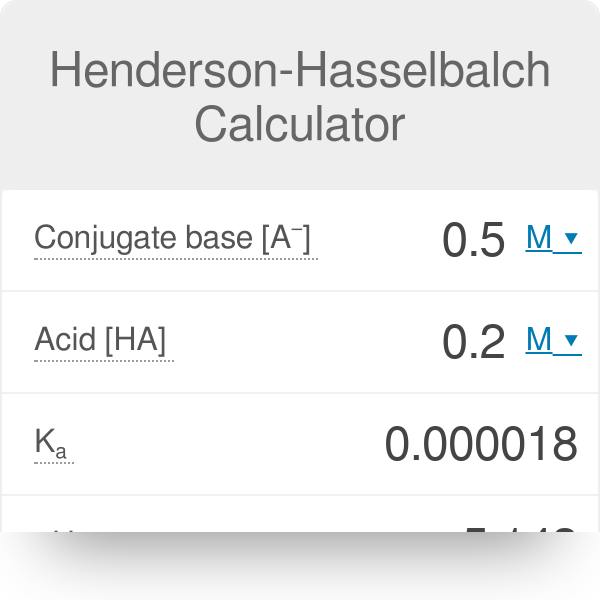

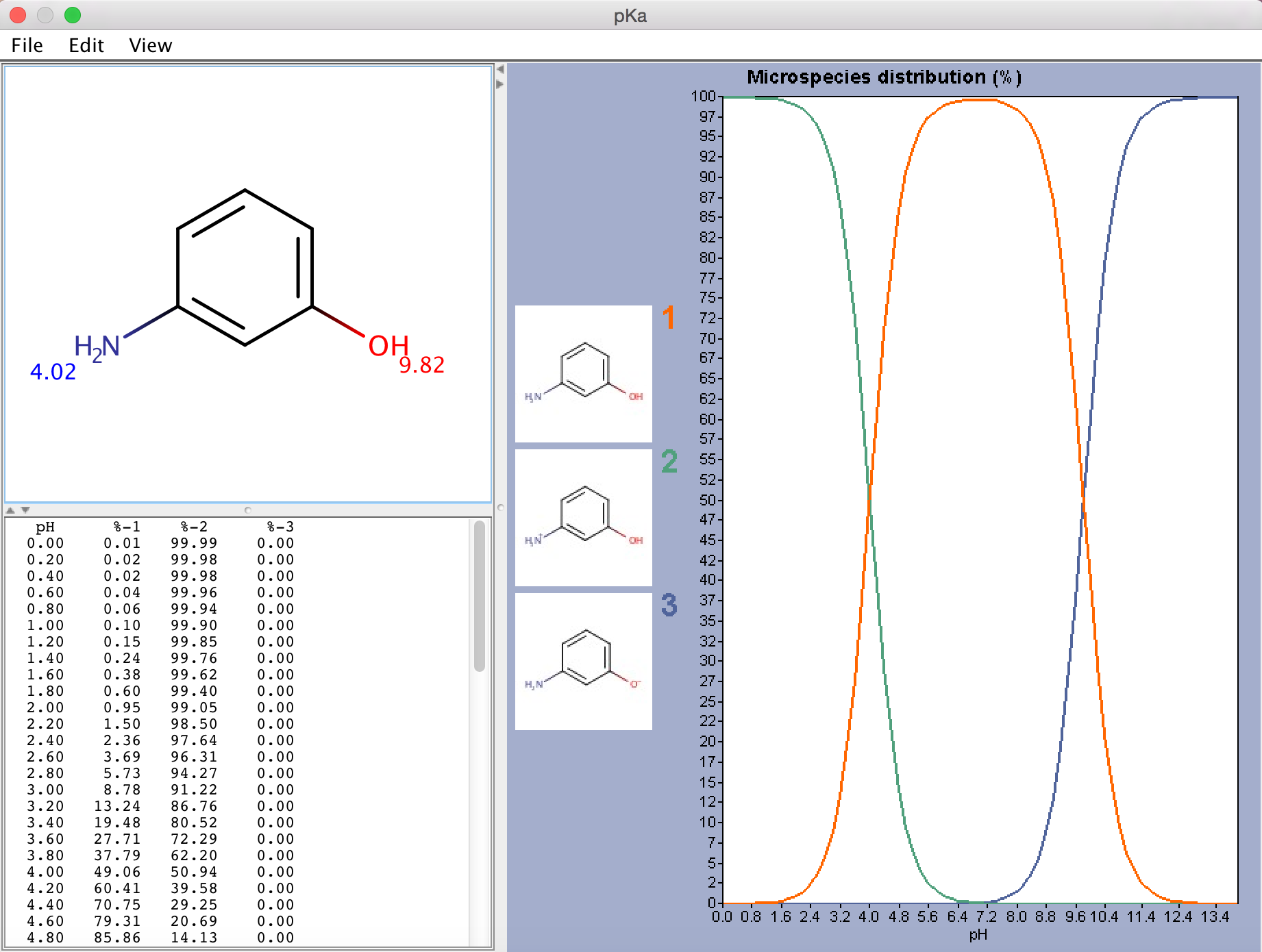
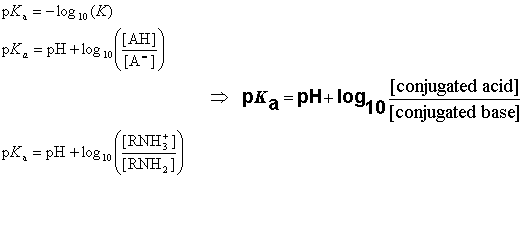
![chemaxoncalcpka [online documentation] chemaxoncalcpka [online documentation]](https://doc.mcule.com/lib/exe/fetch.php?w=800&tok=562d71&media=pka.png)